We've done lot's of scientific exploring so far. We've refined our reasoning, deduction and logic skills. We've slogged through a substantial and unavoidable amount of factual detail, and if you've read some of my other articles, and felt stumped and confused, I feel deep empathy. Know that I too have wanted to hurl my computer out the window many times (between you and me, some of the technical Wiki articles in particular are just awful torture for those of us who don't have the seemingly required Ph. D(s)). The road hasn't always been easy. So today let's load up our science with fun. Gems, to me, are not only fascinating but loaded with fun.
Gemstones, little glittering jewels sprinkled into the largely dull grey/brown matrix we call Earth's crust, beg us to let our imaginations free, to put our scientific tools aside for a moment, and allow their beauty to draw us in. The samples below are just the tip of the iceberg:
Until recently, gems have been an utter mystery to humans. We didn't know where they come from, how they are made, and why they are so stunningly beautiful. For centuries, gemstones have served as tiny vessels allowing mystery and beauty to condense into various restorative and magical powers ready to bestow on us a passageway to magic simply by looking at them.
Rich folklore developed around many gemstones and I urge you to browse some of these fascinating stories. Ellen Steiber provides us with an excellent introduction to gemstone meanings and symbolisms. Mysticalencounter provides a primer and an extensive list of gemstone mystical powers. If you consider our contemporary lore around diamonds and birthstones, you'll recognize some of the powerful symbolic power that still pervades the world of gems today.
Here are a variety of websites perfect for ogling beautiful gems (they have great photos):
Gemselect. Click on each gem to see even more gem photos.
The Mineral and Gemstone Kingdom photo gallery
Alpine Gems, the "Canadian Gemstone" website
Production and Photographs of American Gems
Minerals By Name (An extensive alphabetic list with photographs)
Gyspy Gemstones
Birthstones
John Betts Fine Minerals
Try out a new age crystal shop, pick up and turn over samples in your hands, marvel at all of the intoxicating colours, textures and how they play with the light. Where do they come from? How are they made? And what are they made of?
Browse, relax, set your scientific toolkit aside for a few moments and marvel at each gem's natural magic.
THE SCIENCE
Now that we are in a relaxed place and our chakras are in order, let's ease our way into the science.
Gems, Minerals and Rocks
Gems, minerals, rock - this is the science of geology. Gems and minerals are actually the same thing. But only some minerals earn the name "gemstone." Minerals are naturally occurring substances that are stable solids with specific chemical formulas and ordered atomic structures, and there are a lot of them, about 4300 here on Earth. Minerologists (geologists who focus their study on minerals) utilize a variety of properties such as colour, luster, crystal hardness and transparency in order to identify and characterize them all. What makes a mineral a gemstone is part common sense (hardness, scratch resistance) and part subjective quality- it must have clarity, colour and "fire" - the ability to split light into the colours of the rainbow. The technical word for fire is dispersion, but fire is so much more evocative of the effect. Brilliance, luster and scintillation are other words gemologists use. We will explore these qualities soon. Ultimately what makes a mineral a gem is its beauty.
A mineral is not a rock. Minerals have specific chemical formulas. Rock, however, is a less specific term for all the crust material on Earth. A rock can be an aggregate of different minerals or it may not be made of minerals at all.
To make things even trickier, gems are often minerals but not always. Lapis lazuli is a rock gem that has been mined in Afghanistan since 3000 BC, while amber and jet are organic materials.
Below left is an amazing polished specimen of lapis lazuli.
(fr:User:Luna04; Wikipedia) and (Anders L. Damgaard - www.amber-inclusions.dk;Wikipedia)
Amber, right, is fossilized tree resin, and it has been valued as a gem for over 10,000 years. An ant is perfectly preserved inside this specimen.
Jet, perfectly black, can be polished to a glassy shine. It is composed of carbon
from decaying wood squeezed under enormous pressure. Jet jewelry dates
back to 17,000 BC.
How Gems Are Formed
The Earth's crust is made of several plates that float upon a liquid magma mantle. Where the two layers meet is a zone of enormous pressure and extreme temperatures. The lower surface of the crust, where it meets the mantle churning beneath it, contains many fractures and fluid-filled cavities. These fluids come from mineral-rich magma. As it rises toward the surface during convection, the pressure exerted on it decreases enough to allow gases to escape. These gases can later condense inside rock cavities, creating mineral-rich fluids. Crystals can start to form in the fluid. The cooling of the magma must be very gradual in order for gems to form. Gem-quality crystal formation requires intense heat and pressure, with very gradual cooling. If the magma is too close to the surface, it will cool rapidly and the crystals will be too small. Granite rock will form. It contains small crystal grains of quartz and other minerals that could be the building blocks of gems if they were larger. If the cooling is gradual enough, pegmatite will form instead of granite. It is made of the same minerals but it is much more coarsely grained, with pockets inside it where gemstones with large well-formed crystal structures can form. It is the intense pressure that forces atoms into various tight crystal lattices associated with many gems. This heat and pressure also contributes to the translucency or transparency of many gems. The International Gem Society provides an in-depth explanation of how and where various gems form.
One Formula > Several Gemstones: Corundum Gems As an Example
Many gems (there are over 200 recognized types) come in several different varieties, meaning that one mineral (a single chemical formula) can account for several, often very distinct, gems. For example, sapphires and rubies are both corundum, an aluminum oxide (Al2O3) mineral. Corundum is the hardest of any natural mineral besides diamond. It is often mixed in with grains of beach and river sand (silicon dioxide). It is used as sandpaper grit.
The atoms in corundum, like many but not all gemstones, are organized into tightly fitted together lattices called crystals. Each crystal below is a sample of corundum:
(Ra'ike; Wikipedia)
None of these samples look anything like a ruby or a sapphire. Pure corundum is colourless. It is the inclusion of other elements, usually metals, which give it a range of colours: pink, red, yellow, blue, violet and green.
Rubies and sapphires (and all corundum minerals) have exactly the same crystal structure:
(NIMSoffice; Wikipedia)
What makes some corundum minerals colourful gems is the trace substitution of other metal ions for aluminum ions. Chromium, titanium and iron can all substitute for Al3+, and this substitution is part of the secret of why gems have so many brilliant colours.
left: (Montanbw; Wikipedia) and (Humanfeather:Wikipedia)
 A sapphire, above left, has the same aluminum oxide (Al2O3) mineral structure as the corundum examples. But here, trace amounts of two metal ions - iron (Fe2+ or Fe3+) and titanium (Ti4+), substitute for Al3+ ions in aluminum oxide.
A sapphire, above left, has the same aluminum oxide (Al2O3) mineral structure as the corundum examples. But here, trace amounts of two metal ions - iron (Fe2+ or Fe3+) and titanium (Ti4+), substitute for Al3+ ions in aluminum oxide.A ruby on the other hand, below left, owes its intense red colour to chromium. Like the sapphire, a few Al3+ ions are replaced by other metal ions, in this case chromium (Cr3+).
The Secret of Colour
Our ruby and sapphire examples show us that substitutions with certain metal ions in the crystal structure of corundum give us two very different gems. Aluminum, chromium, titanium and iron are all transition metals.
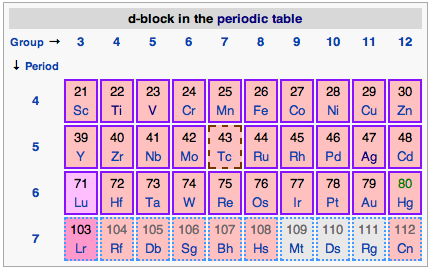
Transition metals, below, are located in the d-block in the periodic table:
Colours and Our Eyes
Before we explore how metal substitutions create brilliant gem colours, let's familiarize ourselves with how our eyes perceive them. The colour of objects is a very complex science in and of itself. For our purposes, I will simplify by saying that the colour of an object tends to be complimentary (opposite) to the colour that its molecules absorb.
This is a colour wheel. Colours on opposite sides of the wheel are complimentary to each other.
(Sakurambo; Wikipedia)
For example, green leaves reflect green light. The green chlorophyll in them absorbs light at both the red end and the blue end of the visible light (sunlight) spectrum, but not green light. The light reflected from the leaf is missing red and blue and therefore it looks green as a result.
How our eyes and brains perceive the colours of various objects is actually very complex, depending on how light reflects or scatters off the object (physical factors), and how our brains interpret the light based on ambient lighting, viewing angle, etc. (an effect called colour constancy).
Some gems even emit light, a phenomenon responsible for the brilliant red intensity of some rubies, as we will see in a moment.
Valence and Colour
Atoms have various energy levels available to their electrons. These are called shells. Atoms always fill up their innermost shells first and move outward from there. The outermost shell contains the electrons that are available for chemical bonding. These are called valence electrons.
The valence number of an element tells you how many covalent bonds that element can form with other atoms. The arrangement of electrons in an atom becomes most stable when the valence shell is either filled or emptied. This is one reason why atoms want to bond with each other - to share electrons and fill their valence shells.
We already saw that iron ions can exist in two different states - Fe2+ and Fe3+. These are called oxidation states. Valence number and oxidation state are the same thing. Some elements have only one valence state, while others like iron have more than one. All the transition metals in the grid above have partially filled valence shells.
Ions containing transition metals, metals with partially filled d valence shells, tend to be coloured.
For example, copper (Cu2+) in solution is light blue. When (an anhydrous - no water - white powder) cupper (II) sulfate is dissolved in water, each copper ion forms an octagonal complex as six water molecules bond with it, along with 2 negative sulfate ions. This happens because water ionizes in solution into a hydroxide (OH-) ion and a hydronium (H30+) ion, as shown here:
Positively charged copper ions will bond with negatively charged hydroxide ions and negatively charged sulfate ions to form a complex. Other transition metal ions in aqueous (water) solution also form various kinds of octagonal arrangements. And when they do, they tend to create energy gaps, which vary depending on the transition metal ion, its oxidation state and the nature of the ligands (the surrounding ions or molecules).
Energy gaps are the key to colour formation. The copper ion has 9 electrons in its outermost (valence) energy shell, all occupying 5 3d orbitals. Electrons in two of the 3d orbitals align with the negative ions as they approach but they also repel them, being negatively charged. This arrangement (of being pushed together) increases the energy of these two 3d orbital electrons. Now, the octagonal complex can absorb a light photon that has an energy equivalent to the energy difference between the electrons in the 2 higher energy 3d orbitals and the remaining 3 regular energy 3d orbital electrons. This phenomenon is called crystal field splitting. The particular energy in the case of this copper/water complex is in the orange region of the spectrum, so orange light is absorbed.
The promotion of each of the three electrons absorbs the amount of energy corresponding to a photon of orange light. The light reflected from the complex (missing orange photons) looks blue as a result, so the Cu2+ solution looks blue to us.
(This is also why non-anhydrous cupper sulfate crystals look blue. These crystals are in a hydrate form, Cu(II) SO4·5H2O. Five water molecules are bound to the copper sulfate.)
This kind of mechanism is especially prevalent in transition metal complexes, which are formed when various gemstones crystallize from aqueous solutions. In order to have colour, the energy gap must be between 1.8 and 3.1 electron volts (eV), because this energy corresponds to the energy of light photons in the visible range, with wavelengths between 400 to 700 nm (nanometers). This range represens very low energy transitions, so they require very closely spaced electron shells, some of which contain electrons and some of which are empty. The partly filled d orbitals of transition metals fit the bill. In most compounds, the energy required for the promotion of an electron requires more than 6 eV. Photons need to be at least in the ultraviolet range to have this much energy. Such compounds are absorbing these UV photons and reflecting light but we can't see it.
Vanadium is another pretty transition metal ion. Dissolved in water, it can achieve one of four different valence states (depending on the solution's pH) of +2 (lilac), +3 (green), +4 (blue) or +5 (yellow), as shown from left to right below:
The cupper and vanadium examples show us how transition metal ions in a crystal can create energy gaps in gemstones that result in vivid colours. Both the sapphire and the ruby exhibit this phenomenon of crystal field splitting to achieve blue and red, respectively. This is not the whole story, however. Colours in gems can be intensified further through various additional mechanisms as well. Again, we will use the sapphire and the ruby as our examples.
Gems Boost Colour Further
Blue sapphires owe their intense blue colour to fairly complex chemistry. A blue boost comes from a transfer of an electron from one metal to another, usually iron (Fe2+ or Fe3+) to titanium (Ti4+), both of which are impurities that substitute for Al3+ ions in the aluminum oxide crystal lattice. This electron transfer creates localized pockets of charge imbalance. It causes a change in the valence states of both iron and titanium. When the iron and titanium ions change valence states, electromagnetic energy is absorbed, specifically yellow light. When an object absorbs yellow light, it looks blue, yellow's complimentary colour, because yellow is missing from the wavelengths reflected from it. It only takes 0.01% titanium and iron impurities in corundum to make the intense blue in a gem-quality blue sapphire.
Like the sapphire, in a ruby, Al3+ ions are replaced by other metal ions, in this case chromium (Cr3+). What is unique here is that each Cr3+ ion is surrounded by six oxygen ions (O2-), creating a crystal arrangement that can achieve two light tricks at the same time, making rubies appear especially brilliant to our appreciative eyes. First, yellow-green light is absorbed by it (through the mechanism of crystal field splitting similar to that of the sapphire). Second, this absorbed light is then re-emitted at a different and specific wavelength as red luminescence. The chromium ions, by absorbing these photons, gain enough energy to emit new photons with a wavelength of exactly 694 nm, pure red light in other words.
Laser technology makes use of this special situation. To make a ruby laser, a tube-shaped artificial ruby is capped on each end by mirrors, one totally silvered and the other half silvered. When light is shone on the tube, chromium atoms in the ruby become excited. Just like above, some of them emit red photons of a specific wavelength. Some of these photons run parallel with the tube so they bounce off the mirrors at each end, stimulating other atoms to emit even more red photons. You end up with a single-phase monochromatic red light beam that eventually leaves through the half silvered mirror, a red laser.
Other Optical Behaviours In Gems
Besides the ability to stun us with vibrant colours, often by using more than one chemical mechanism, gems are ingenious with light. Again, thanks to their tight crystalline chemical structure, they are able to take ambient light and play with it like no other natural material can. Gems are an amusement ride for photons. This light-play is called scintillation.
Refraction
Gems are not flat opaque objects, so not all light is simply absorbed or reflected off their surfaces. Because they often exhibit at least some transparency, much of the light striking the gem will refract into it. This means that the phase velocity of the light traveling from air into the gem at an oblique angle is changed. When that happens, it cause the light beam to change direction. Each gem has its own specific index of refraction. Light entering a gem at its critical angle will achieve total internal reflection. If light intersects with the gem within its critical angle, that facet will act like a mirror and it reflect right off of it.
Gems take light in at every facet (surface) so there is a great deal of refraction going on. Gems with high refractive indexes exhibit more refraction, and because of increased refraction they exhibit greater brilliance and lustre. Diamond, for example, has a refractive index of 2.4, while most glasses have an index of around 1.5. This is why glass cut the same as a diamond won't have nearly as much brilliance. Specific gems are cut at particular facet angles to maximize refraction.
Dispersion
Gems are also prismatic crystals so they can also disperse light into rainbow-like colours. This effect is also due to refraction of white light into its component colours. You can often see this effect on the crown facet (top surface) of gemstones, where you can observe flashes of colours. This is part of the gem's fire. It is most easily observed in light coloured gems, especially diamonds.
Double Refraction
Double refraction also occurs in almost all gemstones, in which incoming light is divided into two rays as it enters the crystal, by two different molecules inside it. You will sometimes see a visual doubling of facets when this occurs. The effect is most notable in zircons:
(Oneworldgemstone; Gemologyproject.com; Wikipedia)
Reflection
Reflection off the hard polished surface of a gem contributes to its external lustre. Light also enters the stone, reflects internally, treating the internal surfaces of the gem like mirrors, and then returns to the eye. This is the gem's internal lustre. Both contribute greatly to the brilliance of the gem.
The tight crystalline structures of gems multiply their beauty by enhancing the various ways that light can play in and off of them. Allaboutgemstones explains all of these plays of light in gems very well.
In the next article we will explore an enormous class of gems called silicate gems. This group includes some of the most beautiful gems available.











No comments:
Post a Comment
Note: Only a member of this blog may post a comment.